Dissolved oxygen (DO) measurements is critical when it comes to industrial applications, as well as environmental monitoring. There are several sensor technologies utilized for DO measurements, with the most common being polarographic, galvanic, and optical sensors. This article reviews the working principle of each of these sensor types, and also compares features among polarographic vs galvanic vs optical DO sensor.
What is Dissolved Oxygen
Dissolved oxygen (DO) is the amount of free, non-compound oxygen that is present in water or any liquid. Utilizing a DO sensor is crucial in the assessment of water quality, pollution control, as well as in the deployment of treatment methods. Generally, sensors interpret DO measurements in two ways, depending on the type. One is DO percent saturation, which refers to the ratio of actual DO content to the maximum possible DO at a specific temperature and pressure. Whereas, DO concentration refers to the actual amount of oxygen in the liquid sample in milligrams per liter (mg/L), or parts per million (ppm).
Polarographic DO Sensor
Polarographic DO sensors consist of a cathode and an anode such as gold and silver, respectively. These are submerged in an electrolyte solution like potassium chloride (KCl), and separated from the sample by an oxygen-permeable membrane.
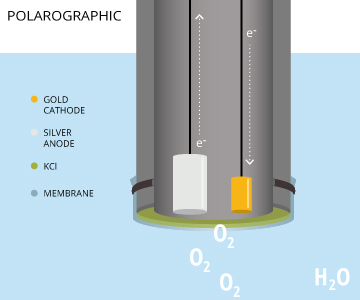
When the instrument is on, the voltage source applies a small voltage (polarization voltage) between the anode and the cathode. Thus, causing oxygen to diffuse through the membrane and undergo a reduction reaction at the cathode.
![]()
The electrons in the above equation are provided by the oxidation reaction occurring at the anode, and electron flow from anode to cathode represents the measuring signal. This signal is proportional to the partial pressure of oxygen in the solution.
Advantages and Disadvantages of Polarographic DO Sensor
Polarographic sensors offer several benefits with a few of them as follows:
- High accuracy and precision.
- Wide measurement range.
- Well-established technology with good track record of reliability.
- Suitable for continuous monitoring.
Despite their advantages, polarographic sensors have some drawbacks:
- Required warm-up time before use.
- Susceptibility to interference from certain gases like hydrogen sulfide, as well as strong acids.
Galvanic DO Sensor
Galvanic DO sensors operate similarly to polarographic sensors, in that they are both electrochemical sensors. However, galvanic sensors generate their own voltage as dissimilar metals (different electro potentials) serve as the anode and cathode. This intrinsic voltage difference between the two metals eliminates the need for an external power source, as well as warm-up time. Usually, the anode is zinc, lead, or another active metal, while the cathode is silver, or another noble metal. The chemical reaction between oxygen and the anode produces a current proportional to the DO concentration.

Galvanic DO Sensor Advantages and Disadvantages
Galvanic sensors offer several benefits:
- Simpler electronics due to self-polarization.
- No warm-up time, so can provide immediate response to oxygen changes.
- Lower power consumption.
Whereas limitations of this sensor type include:
- A shorter lifespan, in general, vs polarographic sensor due to anode consumption.
- Regular membrane and electrolyte replacement required.
Optical DO Sensor
An optical DO sensor has a completely different working principle vs its polarographic and galvanic counterparts. It uses a luminescent dye that fluoresces when exposed to a specific wavelength of light. The presence of oxygen quenches this fluorescence, and the sensor measures the change in fluorescence lifetime or intensity to determine the DO concentration.

Above is a typical optical dissolved oxygen (DO) sensor comprised of several key components. The sensing element contains a luminescent dye that becomes immobilized in a matrix such as sol-gel or xerogel. When the built-in LED emits blue light, the dye reacts to this light. Additionally, some sensors use an LED to emit red light as a reference to enhance accuracy; this red light does not cause luminescence but instead reflects off the dye. The intensity and luminescence lifetime of the dye, when exposed to blue light, are affected by the concentration of dissolved oxygen in the water sample. As oxygen passes through the membrane, it interacts with the dye, which subsequently reduces both the intensity and the lifetime of the luminescence. Finally, a photodetector measures the intensity or lifetime of the returned luminescence, enabling the calculation of dissolved oxygen concentration in the sample.
Advantages and Disadvantages of Optical DO Sensors
Optical sensors offer numerous benefits including:
- Minimal maintenance requirements, so they are ideal for long-term monitoring programs.
- Long-term stability with minimal drift, hence minimizing the need for re-calibration.
- Flow rate independence, so suitable for harsh environments.
- No oxygen consumption during measurements.
Despite these advantages, optical sensors have some drawbacks:
- Higher initial cost of optical DO sensor vs polarographic and galvanic counterparts.
- Slower response time in comparison to electrochemical sensors.
Differences Between Polarographic vs Galvanic vs Optical DO Sensor
All three sensor technologies provide unique features that make one suitable over the other in certain applications. The following table highlights their differences.
| Parameter | Polarographic | Galvanic | Optical |
| Accuracy and Precision | Provides high accuracy across wide measurement range. | Offers good accuracy for a limited measurement range. | Has the highest accuracy and stability. |
| Response Time | Moderate response time, suitable where real-time monitoring is not critical. | Fastest response time, beneficial for dynamic environments requiring immediate data. | Fast response time, ideal for applications needing real-time feedback. |
| Maintenance and Calibration | Regular maintenance and frequent calibration needed to ensure accuracy. | Less maintenance and periodic calibration required. | Minimal maintenance and infrequent calibration, hence reducing downtime. |
| Cost Considerations | Generally cost-effective for laboratory and industrial applications. | Affordable and practical cost for portable and field measurements. | Higher initial cost but lower long-term maintenance costs, offering better value for continuous monitoring applications. |
| Application | Suitable for precise measurements in controlled environments. | Ideal for field applications where portability is essential. | Perfect for continuous monitoring in critical applications, since it is less prone to drift. |
Advanced DO Sensors from Alpha
At Alpha Measurement Solutions, we offer a range of advanced dissolved oxygen sensors that meet the specific needs of different industries. Our offerings include polarographic, galvanic, and optical DO sensors.
Alpha’s Optical Sensor:
- Range: 0 – 20 ppm (0 – 200% saturation) / 0 – 50 ppm (0 – 500% saturation), adjustable within 0 – 50 ppm.
- Response Time: T90 < ~35 seconds.
- Accuracy: 0 to 10 mg/L (±1%), 10 to 20 mg/L (±2%), 20 to 30 mg/L (±5%), 30 to 50 mg/L (±15%).
- Temperature Range: 0 to 50°C.
- Pressure: Operating Atmospheric Pressure: 40 to 115 kPa, Maximum Bearing Pressure: 700 kPa.
- Warranty: 6 to 24 months depending on the solution.
- Features: High stability, minimal drift, and suitable for various environments including biological and hydrocarbon applications.
Alpha’s Polarographic and Galvanic Sensors:
- Range: 0 – 20 ppm (0 – 200% saturation) / 0 – 50 ppm (0 – 500% saturation).
- Response Time: T90 < 35 seconds.
- Accuracy: ±2%.
- Temperature Range: 0 – 50°C continuously monitored, 0 – 80°C grab sample.
- Pressure: Operating Atmospheric Pressure: 40 to 115 kPa, Maximum Bearing Pressure: 700 kPa.
- Warranty: 12 months.
- Features: Various polarographic and galvanic configurations, robust design, suitable for diverse applications including process and field measurements.



