溶解氧(或溶解氧)是指水或任何其他液体中游离的非复合氧的含量。测量 溶解氧 is crucial when assessing water quality, pollution control, and treatment processes. This article reviews current methods of measuring dissolved oxygen, why this measurement is necessary, and the diverse applications that stem from this precise analysis.
Dissolved Oxygen Measurement Methods
溶解氧的测量通常以空气饱和度百分比(% sat)或毫克/升(mg/L)为单位进行监测。此外,还有多种测量方法,有些方法在某些情况下更有优势。
温克勒方法
"(《世界人权宣言》) 温克勒方法 该方法由匈牙利化学家 L.W. Winkler 于 1888 年发明,是最早的方法之一,但至今仍然适用。这是一种滴定法,通过水样中的一系列化学反应来确定溶解氧水平。通常情况下,测量过程需要一个专门的 BOD 瓶,这种瓶子的设计可以使其密封而不会在瓶内滞留空气。测试水样被收集到 BOD 瓶中,然后按比例加入过氧化锰溶液和氢氧化钠溶液等试剂。这一反应会产生氢氧化锰 (II) 沉淀。如果样品中不含水分,沉淀物呈白色,如下图所示。
![]()
但是,如果测试样本中存在溶解氧,它就会发生反应,形成棕色沉淀。
![]()
然后,将棕色沉淀溶解在有碘离子存在的酸中,释放出碘(I2) 根据溶解氧的量来确定。最后,用硫代硫酸钠滴定释放出的碘,以确定氧气的确切含量。
![]()
![]()
Although the Winkler method remains a recognized standard for DO analysis, it introduces the possibilities of human error, sample contamination, inaccuracies, and interferences. There has been development of several modifications to improve accuracy, but titrations remain cumbersome to execute, especially in the field. This is why new technologies focus on quicker and easier calculation methods while maintaining accuracy.
电化学方法
无论是在实验室还是在现场进行测试,使用电化学传感器都是测量溶解氧的常用方法。这些传感器的工作原理是电极表面的氧气消耗,无论是电化学式还是极化式。电化学和极谱溶解氧传感器都是利用电解质溶液中的两个极化电极--阳极和阴极。
A thin, semi-permeable membrane or diaphragm separates the electrodes and electrolyte from the test sample. When taking measurements, the DO diffuses across the diaphragm at a rate proportional to the partial pressure of oxygen in the sample. Then the cathode reduces and consumes the DO, thus producing a directly proportional electric current to the oxygen concentration. This current flows from the cathode to the anode via the ions in the electrolyte and relates to the partial pressure of oxygen as follows:
![]()
等式中的 id,F,Pm(t), A, pO2和 d 分别代表产生的电流、法拉第常数、隔膜的渗透性与温度、阴极表面积、氧分压和隔膜厚度的函数关系。
有必要在样品中搅拌这些传感器,直到溶解氧读数不再增加。这是因为氧分子的消耗导致测量过程依赖于流量。因此,在无流量的情况下,会产生人为的低溶解氧测量值。
电镀传感器
Galvanic sensors will self-polarize after placement in an electrolyte solution. They do not require warm-up time and can operate without externally applied potential. Common material for the anode is zinc or lead, while the cathode is silver or another noble metal. Because the cathode is inert, it does not interfere with the reaction. Instead, it serves only to pass on electrons. Thus, the anode is oxidized, and oxygen is reduced at the surface of the cathode, with the overall reaction as follows:
![]()
这种反应产生的氢氧化锌以白色固体的形式沉淀在传感器的顶端,但不会覆盖或消耗电解液。随着时间的推移,堆积物会变得过多,影响离子在阳极和阴极之间传输电流的能力。此时,有必要更换电解质溶液。
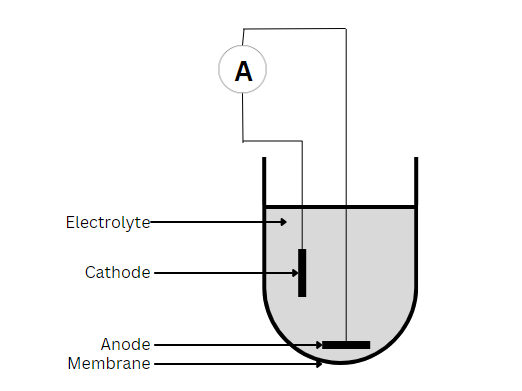
偏振传感器
Polarographic sensors have a setup similar to that of the galvanic sensor but use a silver anode alongside a noble metal like gold or platinum as the cathode. As a result, it needs the application of an external potential (0.4 to 1.2V) to reduce oxygen from the cathode to the anode. Also, most applications require a warm-up period of 5 to 60 minutes. As the silver anode is oxidized, a dark AgCl coating becomes noticeable. However, this occurs only during DO measurements, so the sensor will degrade slowly. This degradation process is a lot slower than that of a galvanic sensor, where the anode precipitates constantly. Over time, as this degradation occurs, the readings will be unstable and unusually low. Hence, necessitating maintenance to restore sensor performance.
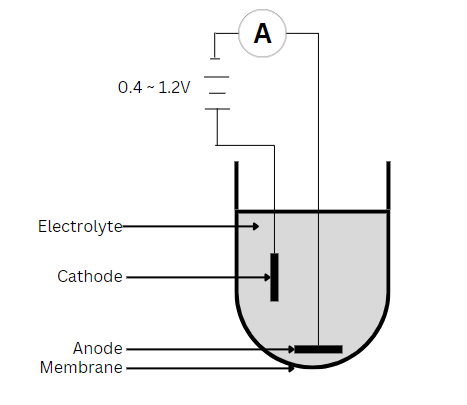
光学方法
Rather than measuring reactions through the electrochemical sensors, optical sensors measure the interactions between dissolved oxygen and certain luminescent dyes. The measurement process begins with exposing the dyes to an incident light. As a result, the molecules of the dyes become excited and will emit light with longer wavelengths as their electrons return to the average energy state. If DO is present, then interactions between the oxygen and dyes will alter and limit the wavelength of the emitted. The frequency, intensity, and decay of the emitted light are related to the partial pressure of oxygen and help to determine its magnitude.
Optical DO sensors require minimum maintenance and are ideal for continuous measurement or long-term applications. Moreover, they are generally more accurate than electrochemical sensors. This is because they are tolerant to hydrogen sulfide or other gasses that can impact the performance of electrochemical sensors. In addition, they are also capable of measuring DO at concentrations much lower than the detection limits of the electrochemical sensors. The significant disadvantages of the optical DO sensors include higher power consumption, slower response time, and accuracy dependent on humidity.
比色法
这种方法需要使用能与溶解氧发生反应的比色试剂,从而产生与溶解氧量成正比的颜色变化。通常使用分光光度计、色度计或简单的比较器来确定颜色变化。比色法的两种常见变体包括靛蓝卡明和 Rhodazine D。
The Indigo Carmine forms a blue-colored solution with DO and can measure its concentrations between 0.2 and 15 ppm. The presence of ions such as ferrous, nitrite, and hydrosulfite interferes with this measurement. Rhodazine D will produce a rose-colored solution with oxygen and can detect much lower DO concentrations at the ppb level. The presence of oxidizing agents such as chlorine, ferric, and cupric will affect its measurement results.
Importance of Dissolved Oxygen Measurement
精确的溶解氧测量是了解水生环境动态健康状况的窗口。如果溶解氧水平较低,则可能表明存在污染、富营养化或微生物活动过多,从而危及水生生物的健康。反之,如果氧气含量较高,则表明存在藻类或曝气装置的影响。
Applications of Dissolved Oxygen Measurement
Dissolved oxygen measurement is common in the following applications:
- 环境监测: DO measurements are fundamental in monitoring the impact of human activities on water bodies. For example, wastewater treatment plants and regulatory bodies rely on these measurements to assess compliance with environmental standards. Measurements are also useful in identifying sources of pollution. Testing DO in these plants helps in understanding the biodegradable organic matter and the biological oxygen demand. Both parameters are indications of the general water quality.
- 水产养殖管理: 保持最佳溶解氧水平对水生生物的健康和生长非常重要。通过持续监测,水产养殖者可以调整通气量和喂食率,防止与氧气有关的压力和死亡。
- 海洋学和湖沼学: 海洋学和湖沼学研究人员利用溶解氧测量来评估营养循环、氧气动态以及气候变化对水生生态系统的影响。这些见解有助于更广泛地了解全球环境动态。



